Awardee of 2023 Joseph Altman Award in Developmental Neuroscience, Tomasz Nowakowski
Tomasz Nowakowski, PhD
Department of Neurological Surgery
Department of Anatomy
Department of Psychiatry and Behavioral Sciences
Weill Institute for Neurosciences,
Eli and Edythe Broad Center for Regeneration Medicine and Stem Cell Research
New York Stem Cell Foundation - Robertson Investigator
Department of Neurological Surgery
Department of Anatomy
Department of Psychiatry and Behavioral Sciences
Weill Institute for Neurosciences,
Eli and Edythe Broad Center for Regeneration Medicine and Stem Cell Research
New York Stem Cell Foundation - Robertson Investigator
University of California San Francisco
I am honored and humbled to have been selected to receive the 2023 Altman Award in Developmental Neuroscience. I am thankful to the Japan Neuroscience Society for their selection, and owe this distinction to the many supportive mentors, colleagues and collaborators, as well members of my laboratory whom I’ve had the privilege to work with over the years.
Dr. Bayer and Dr. Altman have created unprecedented resources for understanding human brain development, including reference atlases of brain anatomy that serve the research community as a one-of-a-kind resource. We depend on their work every day.
As a graduate student at Edinburgh University, I sought to understand molecular mechanisms that regulate the tempo of brain development using mouse as a model system. Specifically, I had focused on the roles of small non-coding RNAs in controlling radial glia differentiation into neurons. Understanding fundamental principles of brain development is critical to identifying points of vulnerability to mutations that underlie neurodevelopmental disorders. Importantly, developing brain tissue is highly heterogeneous, with dynamic processes of radial glia proliferation, neurogenesis, and gliogenesis occurring often concurrently. For my postdoctoral training, I therefore applied emerging technologies that enable whole transcriptome sequencing of single cells to begin to define these developmental trajectories and lineage complexities at the most fundamental level.
My laboratory seeks to unravel the developmental strategies by which radial glia generate the many cell types of the human brain. Over the last five years, we have developed new methods to track individual radial glia and their progeny using lineage tracing. We have unexpectedly found that towards the end of excitatory glutamatergic neurogenesis, human cortical radial glia frequently give rise to cortical inhibitory GABAergic neurons, and such developmental mechanism could explain increased ratio of GABAergic to glutamatergic neurons in the human brain. We have also discovered a new subtype of radial glia which do not contact the pial surface through a basal fiber, but continue to produce cortical astrocytes, and have proposed a model whereby human radial glia undergo lineage bifurcation event that segregates the generation of gray matter and white matter astrocytes. Finally, we have explored a more complex form of heterogeneity, which is regional diversity of radial glia lineages across the prospective areas of the cerebral cortex. We discovered that epigenomic information encoded in chromatin state present in neural progenitor cells precede transcriptomic distinctions between area specific glutamatergic neurons. Together, our studies have laid the foundation for understanding how genetic, cellular, and intercellular mechanisms organize developing brain. Inspired by Dr. Altman and Dr. Bayer who created anatomical reference atlas of the human brain, my goal is to create a comprehensive blueprint of molecular neuroanatomy of normal human brain development that will serve as a foundation for studying the consequences of genetic and environmental perturbations on neurodevelopmental processes.
My mission is to advance the understanding of human brain development and its relationship to neurodevelopmental, behavioral, and intellectual disorders through basic science research. Central to this goal is training the next generation of scientist, and I have had an incredible privilege to work with many outstanding graduate and postdoctoral trainees who have been advancing the field through their research.
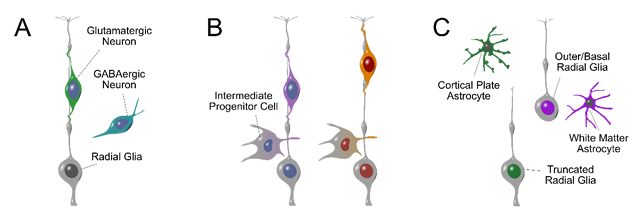
Figure legend:
Developmental strategies of neural stem cells. A) Human radial glia can generate excitatory glutamatergic and inhibitory GABAergic neurons (Delgado et al. 2022, PMID: 34912114). B) Glutamatergic neurons born in different cortical areas follow distinct molecular trajectories (shown as blue/purple or burgundy/orange colors). Limited distinctions in radial glia and neural progenitor cells cascade into robust transcriptomic distinctions in glutamatergic neurons (Ziffra et al. 2021, PMID: 34616060). C) Diversification of radial glia into truncated and outer/basal subtypes correlate with differences in astrocyte differentiation (Allen et al. 2022, PMID: 35587512).

Tomasz Nowakowski, PhD
Educational background
2004-2007:
BSc (Hons.) Biological Sciences (Physiology) - The University of Edinburgh
2007-2008:
MSc (Res.) Life Sciences - The University of Edinburgh
2008-2012:
PhD Wellcome Trust Program in Molecular and Cellular Basis of Disease - The University of Edinburgh
Work experience
2012-2017:
Postdoctoral Fellow, University of California San Francisco, San Francisco, California, USA
2017-2023:
Assistant Professor, University of California San Francisco, San Francisco, California, USA
2023-:
Associate Professor, University of California San Francisco, San Francisco, California, USA




